L. Invernizzi, C. Damiano, E. Gallo
Chem. Eur. J. 2025, 0, e202500473
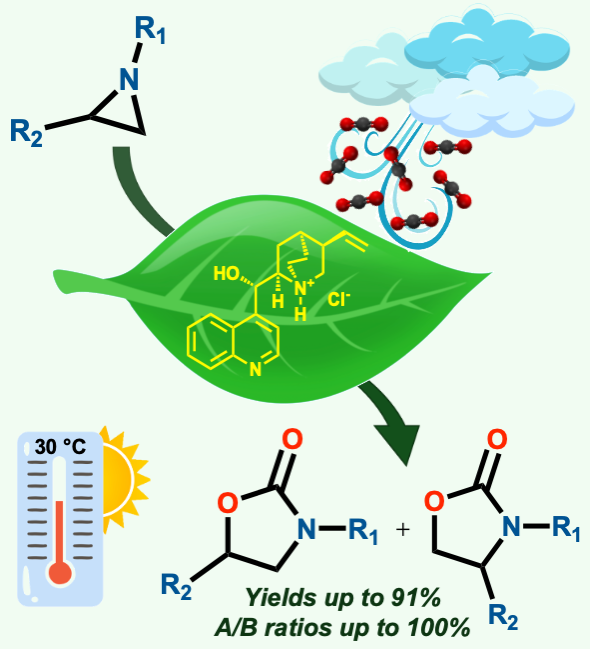
A metal-free, biocompatible catalyst for the cycloaddition of CO2 to N-alkyl aziridines was easily obtained by protonating the natural and nontoxic alkaloid (+)-cinchonine. This bifunctional catalytic system promoted the synthesis of the desired products under very mild experimental conditions (room temperature and atmospheric CO2 pressure) and without the aid of any cocatalyst. No specific equipment is required, making the procedure practical for application in any laboratory. The high synthetic value of this methodology can be attributed to the combination of excellent regioselectivity in oxazolidinone synthesis and the remarkable chemical stability of the catalyst, which can be recycled and reused for at least three consecutive cycles without any significant loss of activity.